Mastering fibrosis through targeted therapeutics
Fibrosis is a pathological wound healing process in which connective tissue replaces normal parenchymal tissue.
As the process continues unchecked, this leads to considerable tissue remodelling and deposition of connective tissue.
The result is interference or complete inhibition and loss of function of the organ affected.
There are no approved drugs which effectively inhibit progression of tissue fibrosis for any chronic systemic disease.
The fibrosis mechanism contributes to life-limiting diseases.
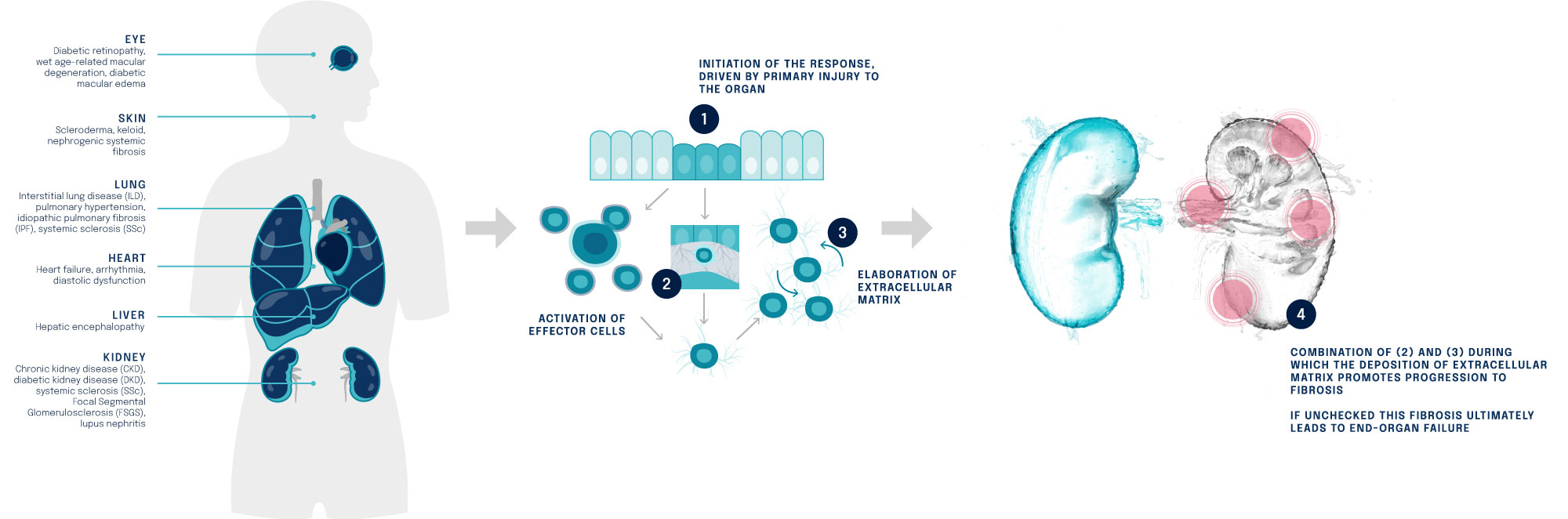
Figure: Fibrosis is a highly dynamic process. It contributes to many life-limiting fibrotic diseases including Chronic Kidney Disease (CKD), Systemic Sclerosis (SSc), Focal Segmental Glomerulosclerosis (FSGS) and others.
Our research has identified specific small molecule inhibitors against a G protein-coupled receptor, a previously undrugged and novel biologic target that regulated fibrosis.
G protein-coupled receptor 68 (GPR68) is silent in healthy tissue but activated following injury or disease. GPR68 has the potential to be a master switch of fibrosis that regulates multiple downstream pathways causing inflammation and fibrosis.
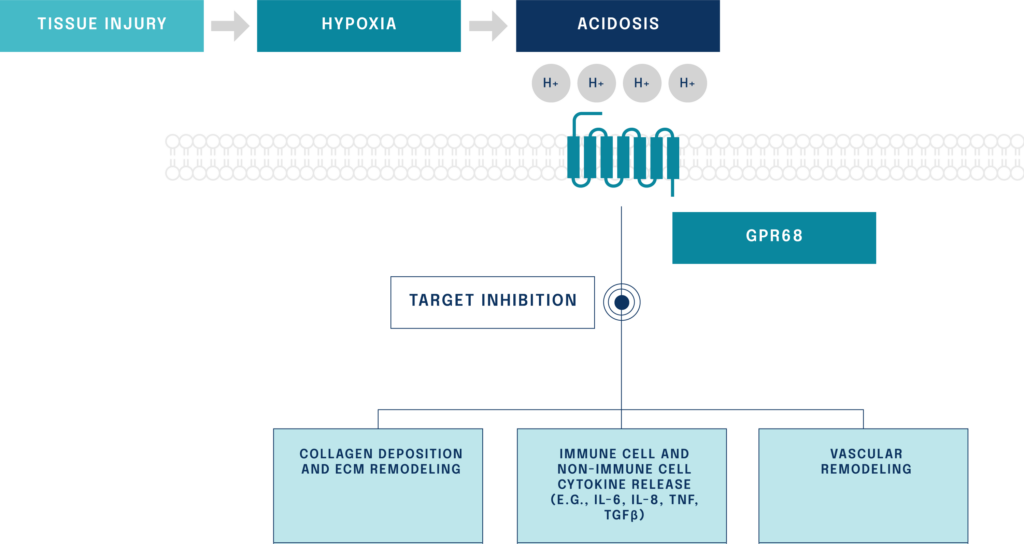
Figure: Inhibition of the novel target GPR68 affects multiple pathways post-injury
GPR68 is a mediator of inflammatory and fibrotic pathways
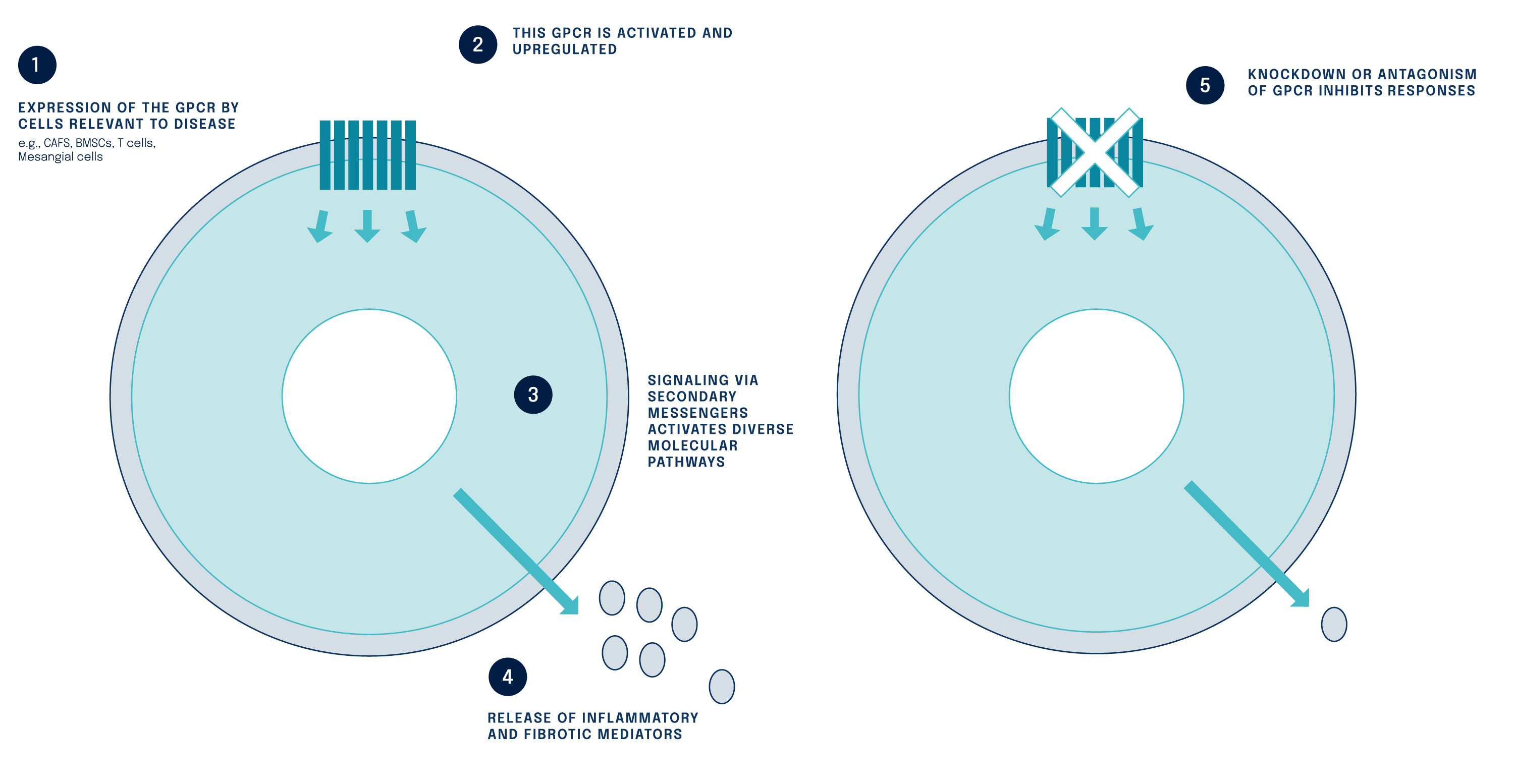
Figure: The fibrosis mechanism of action has been characterised
GPR68 has historically proven challenging to drug but based on decades of scientific understanding and drug discovery, Certa has assembled a pipeline of GPR68 inhibitors (antagonists) tailored for different fibrotic indications.
The pipeline has been developed by enhancing known chemistry to produce novel chemical series with improved activity and tailored for specific indications.
Pre-clinical and early clinical trial data demonstrates the role of GPR68 in regulating multiple downstream pathways that lead to inflammation and fibrosis.
The company is seeking to combine its innovative therapeutics with biomarkers and genetic analysis to identify patients most likely to benefit from treatment, potentially providing a precision therapy.
Developing innovative precision therapies for life limiting fibrotic diseases
Having identified and validated the role of GPR68, a G protein-coupled receptor (GPCR) receptor, as a potential master switch regulating fibrosis. The company has built a platform to discover candidate drugs as potential treatments for multiple fibrotic diseases including serious and chronic conditions impacting the kidney, lung, skin, and heart.
Find more on some of these fibrotic diseases below.
Scleroderma
Scleroderma: a rare and potentially life-threatening autoimmune disorder characterised by inflammation and fibrosis of the skin and other organs.
Chronic Kidney Disease
Chronic kidney disease (CKD) encompasses all degrees of decreased kidney function, from mild or moderate disease to severe and end organ failure.
Focal Segmental Glomerulosclerosis
Focal segmental glomerulosclerosis is one of the many kidney conditions described under the umbrella of glomerulonephritis (inflammation of the kidney), which can lead to chronic kidney disease. FSGS is a condition that results in hardening or thickening of the glomeruli, which act as filters inside the nephrons of the kidney.
Diabetic Nephropathy
Diabetic nephropathy, or diabetic kidney disease (DKD) is a common complication of diabetes, where there is a decline in kidney function and reduced ability to filter out excess proteins from the blood.
Diabetic Retinopathy
Fibrosis is observed in multiple eye diseases, including diabetic retinopathy (DR) and age-related macular degeneration (AMD), where the deposition of scar tissue is irreversible, impacting vision and, in the case of DR, can ultimately lead to blindness.
Disclaimer
All of the candidate drugs described on this site are investigational products which have not received marketing authorization or approval by any regulatory agency, including the US Food and Drug Administration, the European Medicines Agency, or the Australian Therapeutic Goods Agency. The investigational drug products are undergoing clinical studies to evaluate the safety and effectiveness in humans.