Diabetic
Retinopathy
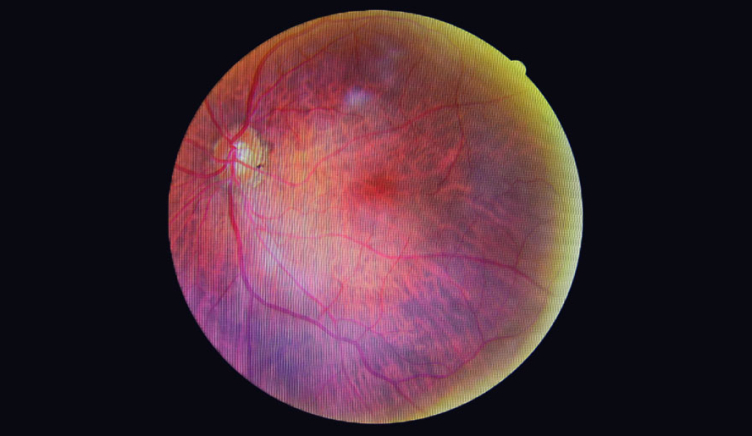
Patients with diabetes often develop ophthalmic complications, such as corneal abnormalities, glaucoma, iris neovascularization, cataracts, and neuropathies. The most common and potentially most blinding of these complications, however, is diabetic retinopathy (DR). DR is a progressive eye disease that worsens over time as the result of damage to the blood vessels of the retina at the back of the eye.
DR affects almost all individuals with type 1 diabetes and more than 60% of those with type 2 diabetes within 20 years of living with diabetes.
DR is the leading cause of new blindness in persons aged 25-74 years in the United States. Approximately 700,000 persons in the United States have proliferative diabetic retinopathy, with an annual incidence of 65,000. An estimate of the prevalence of diabetic retinopathy in the United States showed a high prevalence of 28.5% among those with diabetes aged 40 years or older. Diabetes-related blindness is a personal catastrophe to the individual and costs the United States approximately $500 million annually.
DR is the consequence of chronic progressive diabetic microvascular leakage and occlusion in the retina. It eventually occurs to some degree in all patients with diabetes mellitus. There are two types of DR: non-proliferative and proliferative disease.
Non-proliferative diabetic retinopathy (NPDR) is the early stage of the disease and is less severe. Blood vessels in the eye may leak fluid into the retina, which leads to blurred vision. Proliferative diabetic retinopathy (PDR) is the more advanced form of the disease. New blood vessels start to grow in the eye (neovascularisation), which are fragile and can haemorrhage. Recruitment and activation of inflammatory cells from the circulation can lead to severe scar tissue formation in the retina and permanent vision loss.
Treatment of DR includes intravitreal administration of corticosteroids or monoclonal antibody products in addition to laser therapy or retinal coagulation.
There are currently no treatment options that act on the scar tissue formation and fibrosis in the retina, hence a substantial need for therapies to prevent the severe vision loss and blindness in people living with diabetic retinopathy.
Disclaimer
All of the candidate drugs described on this site are investigational products which have not received marketing authorization or approval by any regulatory agency, including the US Food and Drug Administration, the European Medicines Agency, or the Australian Therapeutic Goods Agency. The investigational drug products are undergoing clinical studies to evaluate the safety and effectiveness in humans.